Abstract
Background: Immune thrombocytopenia(ITP) is a complex autoimmune disease characterized by an isolated thrombocytopenia. It has long been known that B cells produce autoantibodies that are directed against various platelet membrane receptors, eventually resulting in platelet destruction by the reticuloendothelial system. T cell has recently been shown to play a critical role in ITP pathogenesis. In addition to platelet destruction, platelet production can be decreased by an immune-mediated megakaryocyte maturation abnormality and abnormal apoptosis. Despite all this, the pathogenesis of childhood ITP has been proven complex, better knowledge of the childhood ITP pathogenesis continues to be a major objective for the near future. And to our knowledge, there is no specialized gene expression study by means of RNA sequencing (RNA-seq) to characterize the whole transcriptome change of childhood ITP. Thus, we performed RNA-Seq of two childhood ITP patients to identify differentially expressed genes and key biological processes involved in childhood ITP.
Methods: Mononuclear cells were separated from the bone marrow samples of two childhood ITP patients. Then we used bioinformatics methods to identify statistically differentially expressed genes and performed functional enrichment analysis to characterize the important functions or signal pathways. To investigate the change of whole transcriptome between ITP and control group and identify potential new functions associated with ITP pathogenesis, we performed Gene Set Enrichment Analysis (GSEA) through multiple functional databases including GO, KEGG and Reactome to determine statistically significant functional gene sets.
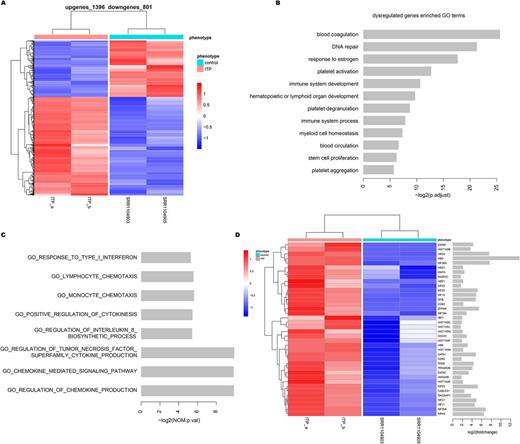
Results: We obtained more than 16 million paired-end reads in two ITP patients. Each sample could well map to reference genome with average 85.31% rate of unique mapping reads. By comparing the differences of transcriptome between ITP and control group, we identified 2197 significantly differentially expressed genes, including 1396 up-regulated genes and 801 down-regulated genes (Figure A). Among these genes, we found 12 genes were not only known disease genes of ITP but also significantly differentially expressed between ITP and control group, such as FOXP3, HES1 and ITGA2B. These differentially expressed genes were involved in GO functions associated with immune, blood coagulation and platelet development, for example, platelet aggregation, platelet degranulation and immune system development (Figure B). Moreover, these genes were also significantly involved in KEGG pathways, such as oxidative phosphorylation and cell cycle pathway.
To analyze the function impact of B cells and T cells in ITP patients, we performed GSEA for gene expression profile. The results showed that genes were significantly enriched with B cell and T cell-mediated immunity and the core enrichment genes were clearly up-regulation in ITP patients. In addition, the differentially expressed genes between ITP and control group were enriched in functions related with cytokines (Figure C). By comparing the differential expression genes with known genes encoding cytokines, 9 cytokine genes were significantly differentially expressed between ITP patients and controls, such as CXCL12, BMP3 and IL-24. Further more, we found that the gene expression profile was positively enriched with genes involved in megakaryocyte development and platelet production (Figure D). 5 megakaryopoiesis-associated genes showed significantly abnormal expression, including GATA1, MYB, CXCL12, ANGPT2 and COL1A1. Last but not least, several potential new pathways were also discovered, such as ECM receptor interaction, cell cycle, oxidative phosphorylation, focal adhesion and leukocyte transendothelial migration.
Conclusions: Our results characterized the whole transcriptome change of childhood ITP patients and elucidated the genes and pathways consistently aberrant in ITP. These abnormal expressed genes, cytokines and pathways may play important roles in the pathogenesis of childhood ITP patients, and may serve as potential targets of diagnosis or treatment.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.